Precision You Can Measure. Purity You Can Trust.
In research, variables are the enemy. That’s why we’ve built our entire operation around delivering peptides with verified purity, accurate dosing, and documented safety. Your results depend on it. So do we.
TelegramThe Three Pillars of Our Quality Standard
Safety for Bioassays
Endotoxins and bioburden can kill cell lines or trigger immune responses, ruining your assay. We test to ensure our products are free from microbial contamination and harmful endotoxins, protecting the integrity of your in vivo and in vitro work.
Exact Potency & Mass
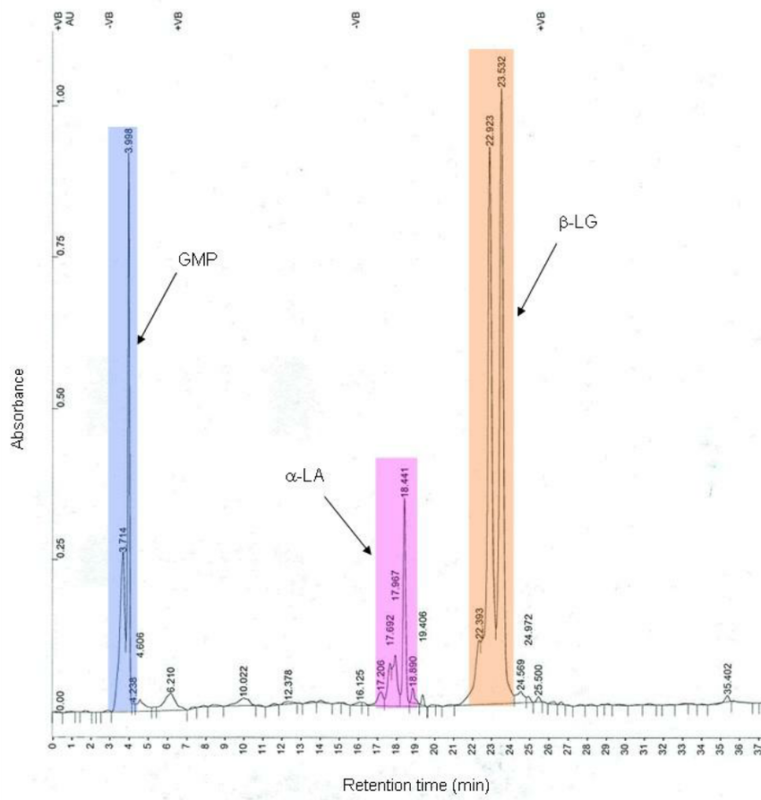
“Under-dosed” or “over-dosed” products lead to irreproducible data. We use Mass Spectrometry to confirm the precise molecular weight and identity of every batch, ensuring you get the exact concentration stated on the vial.
Verified Purity (>99%)
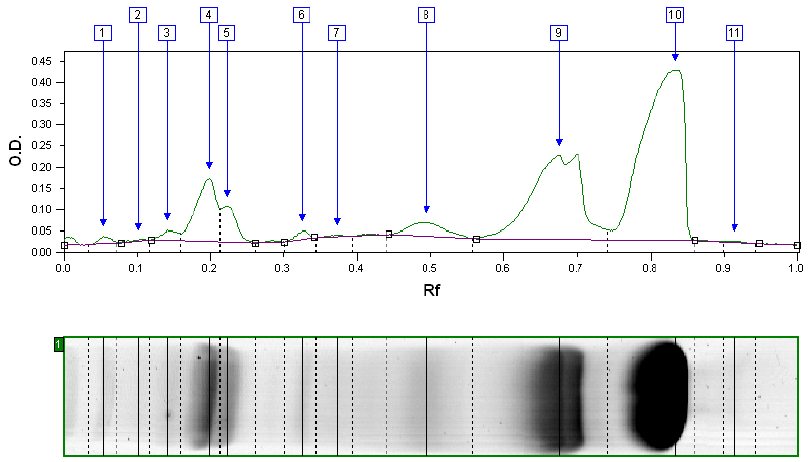
Impurities can act as confounding variables, rendering your research invalid. We utilize advanced purification methods to ensure our peptides meet or exceed 99% purity, giving you a clean baseline for your experiments.
From Synthesis to Your Lab
A Transparent Journey: How Quality Reaches You.
GMP-Compliant Synthesis
Our manufacturing partners adhere to Good Manufacturing Practices (GMP), using pharmaceutical-grade reagents in controlled cleanroom environments (ISO Class 7 or better).
Purification & Analysis
Raw peptides undergo High-Performance Liquid Chromatography (HPLC) for purification, immediately followed by in-house analytical testing for initial purity verification.
Independent Validation
Representative samples are sent to a third-party, accredited lab for confirmatory testing: HPLC for purity, Mass Spec for identity, and LAL testing for endotoxins.
Secure Packaging & Shipping
Once verified, peptides are vialed in sterile, medical-grade glass, sealed, and shipped as a stable, lyophilized powder to preserve integrity until you are ready for reconstitution.
Proof in the Data: The Certificate of Analysis (CoA)
We Don’t Just Claim Purity. We Prove It.
A Certificate of Analysis (CoA) is the birth certificate for your product. It’s a document from an accredited laboratory that provides the raw data on what’s actually in the vial. For us, a product isn’t ready to ship until this data says it is.